|
The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39. Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. That’s the total molar mass of the compound CO2. 
Then finally you add all products, so 12 plus 32 is 44. You would multiply it by its subscript of 2, which is 32. Then you’d do the same thing with oxygen. Enter a chemical formula to calculate its molar mass (e.g. Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44. Then, to find the molar mass of the compound, you’re going to add all those products together. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. 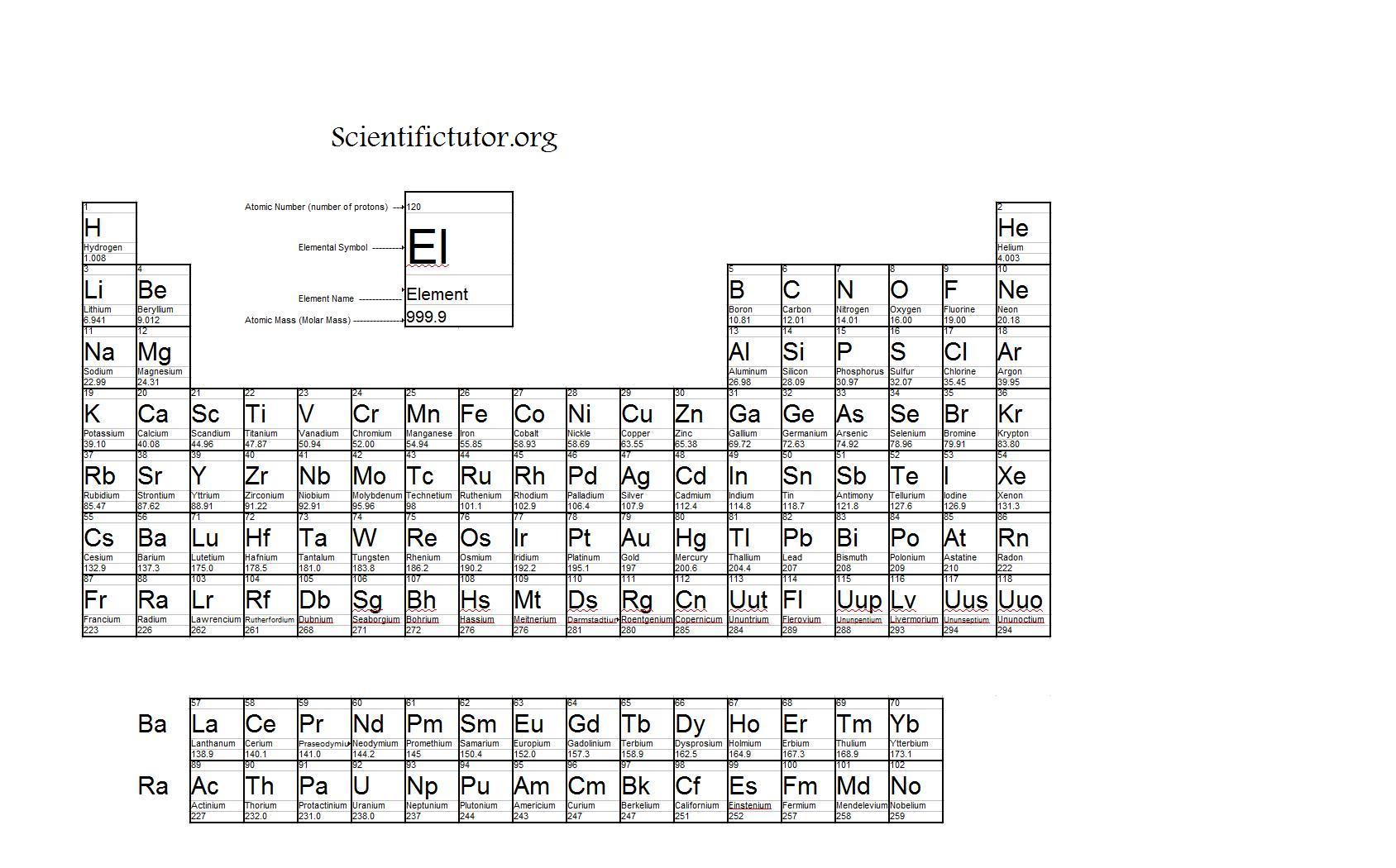
After you’ve found the atomic mass, multiply the atomic mass by the subscript. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. That’s how you find the molar mass of an element. Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\). If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table. 
To calculate the molar mass of a substance you need a periodic table and the chemical formula. Find the atomic mass for each element using the mass shown in the Periodic Table or Atomic Weight Table. The mass in g of 1 mole of a substance is known as the molar mass or molecular weight of the substance. The mass of a mole of substance is called the molar mass. Molar Mass (Molecular Weight) - The term mole also referred to as mol was first used by Ostwald in 1896.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
